 
These features are accompanied by the production of water as only reaction product, which adds further advantages from the sustainability point of view. Indeed, the chemical to electric energy conversion may reach 60% by itself, while increasing efficiency is achieved when recovering also heat in Combined Heat & Power (CHP) co-generation plants. An alternative use is as fuel for fuel cells (FCs), electrochemical devices that are characterised by higher efficiency than internal combustion engines. Its direct combustion, though energy efficient, is difficult due to hard thermal management for the very high adiabatic temperature raise, which imposes the selection of appropriate materials for burners and the control of NO x emission, when combustion is carried out with air. 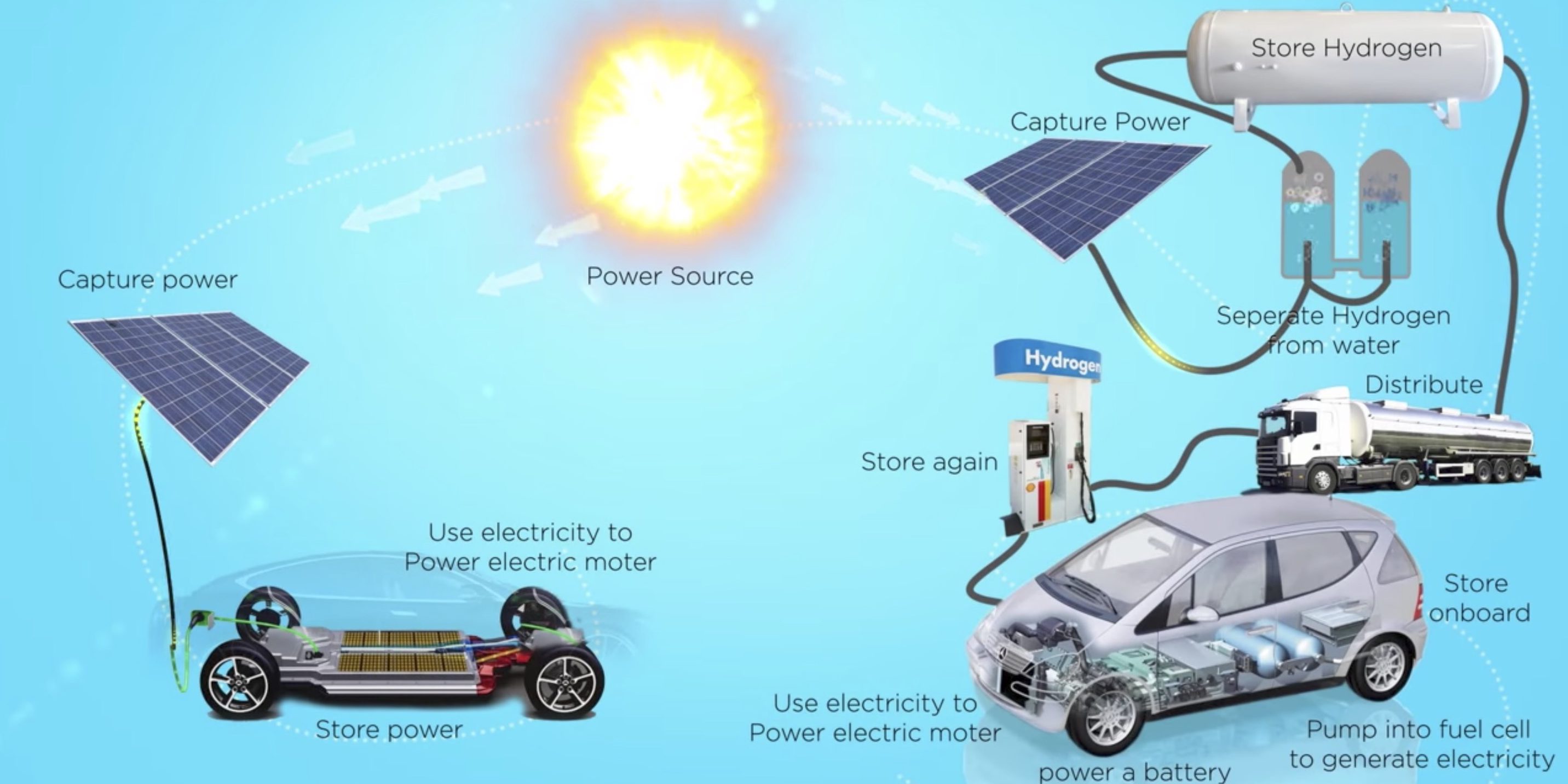
Hydrogen is the most abundant element in the universe and raised particular interest in recent times due to its high energy content per unit mass: its enthalpy of combustion is − 286 kJ/mol. The economic feasibility seems demonstrated when producing hydrogen starting from diluted bioethanol. Examples of steady state or dynamic simulation of a centralised or distributed H 2 production unit are presented to demonstrate the feasibility of this technology, that appears as one of the nearest to market. The case history of hydrogen production from bioethanol for use in fuel cells is detailed from the point of view of process design and techno-economic validation. for the H 2 production from reforming, aqueous phase reforming, biomass pyrolysis and gasification, photo- and electro-catalytic processes, enzymatic catalysis. Selected examples are described, highlighting whenever possible the role of catalysis and the open issues, e.g. In every case, for the main processes that reached a sufficiently mature development stage, attempts of process design, economic and environmental impact assessment are presented, on one hand to finalise the demonstration of the technology, on the other hand to highlight the challenges and bottlenecks. Catalysts are directly involved in the main transformation, as in the case of reforming and of electro-/photo-catalytic water splitting, or in the upgrade and refining of the main reaction products, as in the case of tar reforming. Catalytic processes are used in most options for the production of hydrogen from renewable sources. In addition, the possibility to produce more sustainable hydrogen for industrial application is also of interest for fundamental industrial processes, such as ammonia and methanol synthesis. Hydrogen production from renewable sources is gaining increasing importance for application as fuel, in particular with high efficiency and low impact devices such as fuel cells. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
